Archive: 2026/03
- March
31
2026 - 5
Post-Transplant Infections: Prevention, Vaccines, and Monitoring
Navigate life after organ transplant with practical strategies for infection prevention, vaccination timing, and essential monitoring protocols to protect your immune health.
Read More- March
31
2026 - 5
China vs India Pharmaceutical Manufacturing: Risks, FDA Monitoring & Quality Control
Explore comparative risks in pharmaceutical manufacturing between China and India. Analyze FDA inspection rates, regulatory compliance profiles, and supply chain dependencies.
Read More- March
27
2026 - 5
Individual Variation in Drug Side Effects: Why Medications Affect People Differently
Explore why medications affect individuals differently due to genetics and biology, learning how pharmacogenomics prevents adverse drug reactions.
Read More- March
24
2026 - 5
Medical Alert Bracelets: When and Why They Matter for Drug Safety
Medical alert bracelets can prevent deadly drug errors in emergencies by clearly listing allergies, critical medications, and conditions. They're not optional-they're essential for anyone on blood thinners, insulin, or with severe allergies.
Read More- March
20
2026 - 5
NSAIDs and Peptic Ulcer Disease: Understanding the Risk of Gastrointestinal Bleeding
NSAIDs help with pain and inflammation but carry a serious risk of gastrointestinal bleeding, especially in older adults and those with other health conditions. Learn who's at risk and how to protect yourself.
Read More- March
17
2026 - 5
Intranasal Corticosteroids vs Antihistamines: What Works Better and When to Use Each
Intranasal corticosteroids are more effective than antihistamines for nasal allergy symptoms, especially when used as needed. Learn why they're now the recommended first-line treatment and how to use them properly.
Read More- March
16
2026 - 5
Regulatory Oversight of Online Pharmacies: FDA and State Roles Explained
Learn how the FDA and state pharmacy boards work together to regulate online pharmacies, prevent fake drugs, and protect patients from dangerous websites. Know what to look for and how to stay safe.
Read More- March
13
2026 - 5
Manufacturing Changes and Generic Approval: What Triggers FDA Re-Evaluation
Manufacturing changes in generic drugs can trigger FDA re-evaluation if they affect safety, quality, or bioequivalence. Learn what changes require approval, how long reviews take, and how new FDA programs are speeding up the process.
Read More- March
10
2026 - 5
Opioid Rotation: How Switching Medications Reduces Side Effects
Opioid rotation helps patients with chronic pain switch to a different opioid to reduce side effects like nausea, drowsiness, or confusion - without increasing the dose. It’s a proven strategy backed by clinical evidence and expert guidelines.
Read More- March
9
2026 - 5
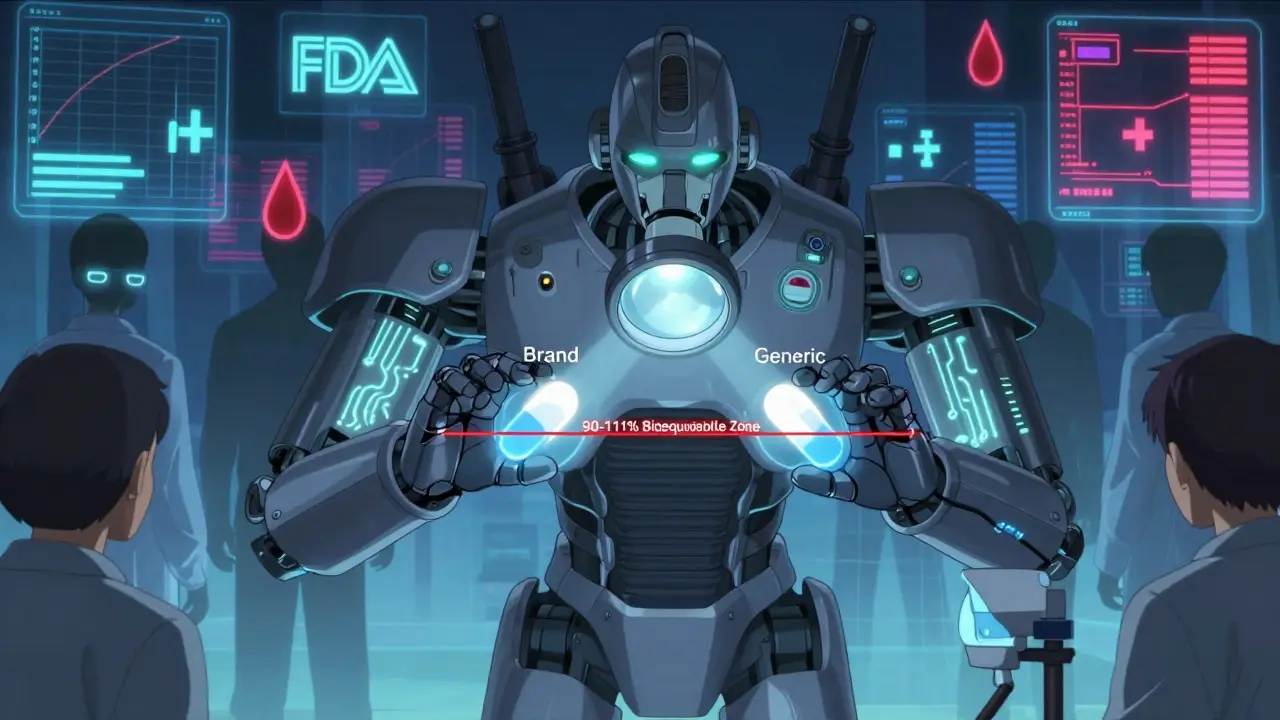
FDA Bioequivalence Standards for NTI Drugs: Special Requirements Explained
The FDA requires stricter bioequivalence standards for NTI drugs like warfarin, digoxin, and phenytoin, with tighter limits (90-111%) and scaled testing to ensure patient safety. These rules prevent dangerous variations in generic versions.
Read More- March
6
2026 - 5
Rheumatoid Arthritis: How Biologic DMARDs Can Lead to Disease Remission
Biologic DMARDs have transformed rheumatoid arthritis treatment, helping 20-50% of patients achieve remission. Learn how these targeted therapies work, which ones are most effective, and what to expect from treatment.
Read More